Chemical Characterization for Medical Devices
Discover the benefits of adding Chemical Characterization for Medical Devices to your overall Safety and Biocompatibility Evaluation Plan of your device.
Nelson Labs
MEDDEV 2.7.1 Rev 4: New Requirements and Changes for Clinical Evaluation Reports (CER)
In June 2016, the MEDDEV 2.7.1 rev 4, the European guidance on the clinical evaluation of medical devices (MD) was published. The issuance of this guidance ...
GMED
Biocomaptibility and the New MDR
This presentation will include an overview of biocompatibility in compliance with the new MDR. The key focus will be on highlighting differences on previous ...
Nelson Labs
TOX21: A New Way to Evaluate Chemical Safety and assess risk
How do you know if the chemicals in the stuff you use and eat are safe? Only a small fraction of the thousands of chemicals in use today have been thoroughly ...
Risk Bites
The New ISO 10993-18 & Updates to Regulatory Expectations Regarding Chemistry
The basic theory of how medical devices should be evaluated for biocompatibility has been in a period of flux. A cornerstone of the new ISO 10993-1:2018 is that ...
Nelson Labs
Satisfying ISO 18562 & FDA Biocompatibility Regulatory Requirements for Breathing Gas Pathway
In March of 2017, the new ISO 18562 standard series was released. This four part standard covers the general principles regarding biocompatibility assessment ...
Nelson Labs
Guidance for Cleaning, Disinfection and Sterilization of Reusable Medical Devices
Presenters: Stefanie Roberfroid and Melissa Galle All reusable devices should contain specific instructions for cleaning, disinfecting and/or sterilization of the ...
Nelson Labs
Biocompatibility of raw materials for medical devices
Starting with a biocompatible material is important for medical device manufacturers. However, regulation is pushing the manufacturer to ask for more ...
Nelson Labs
Drug Discovery Phases = Introduction to Drug Development (HINDI) By Solution Pharmacy
Important Link- Experimental Animals used in Pharmacology Laboratory- https://www.youtube.com/watch?v=kAxTb... Hot Plate Method- ...
Solution- Pharmacy
Chemical Safety
This video describes the risks associated with chemical agents, various types of hazard communications and safe storage of chemicals.
LabsforLifeProject
What are the changes to ISO 14971 2019? (REPLAY) #medicaldevice
ISO 14971:2019 is one of the big standards used by medical device companies to build their Risk Management System. This is so well known that we think it is ...
Easy Medical Device
Using Neuroscience to Evaluate and Guide Treatment for Pediatric Mood Disorders
Each month The Brain & Behavior Research Foundation hosts a Meet the Scientist Webinar featuring a researcher discussing the latest findings related to ...
Brain & Behavior Research Foundation
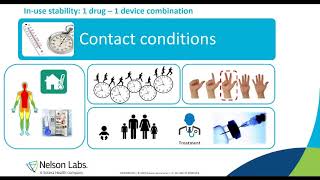
When Drug Meets Device
Presenters: Ruth Verplaetse PhD & Lise Vanderkelen PhD The frequent contact of patients with combined products, a concept that describes the joint use of ...
Nelson Labs
How to Use Biocompatibility to Evaluate Changes in a Medical Device
Medical devices often go through many changes throughout their normal product lifecycles. Whenever a device goes through one of these changes, it should ...
Nelson Labs
What means Sufficient Clinical Data with Bassil Akra (EU MDR)
Webpage: https://podcast.easymedicaldevice.com/78/ You are maybe trying to build your Clinical Evaluation Report or your PMCF for a Medical Device. And you ...
Easy Medical Device
Biosafety Risk Assessment Fundamentals for Clinical Laboratories
Presented at: Clinical Diagnostics & Research 2017 Presented by: Michael Pentella, PhD - Clinical Professor at the University of Iowa, College of Public Health ...
LabRoots
The Power of Chemical Characterization to Assess Changes in Your Medical Device
Tech Theater: The Power of Chemical Characterization to Assess Changes in Your Medical Device 1:30 pm – 2:15 pm This presentation will discuss chemical ...
Nelson Labs
What Manufacturers Need to Know about the Updated ISO 10993-1 and New ISO 21726
The approach expected by regulators for evaluation of medical device biocompatibility has been rapidly changing as expectations within regulatory bodies has ...
Nelson Labs
Medical Devices Regulation Training
MedTech Europe's training on Medical Devices Regulation.
MedTechEurope
Early in vivo Safety Toxicology Considerations
Brian Roche, PhD, Director of Safety Pharmacology at Charles River Laboratories, Inc., presents at the Drug Development "Boot Camp": Practical Aspects of ...
Ohio State University Comprehensive Cancer Center-James Cancer Hospital & Solove Research Institute
Poisoning and Toxidromes: Definitions, Types & Diagnosis – Emergency Medicine | Lecturio
This video “Poisoning and Toxidromes: Definitions, Types & Diagnosis” is part of the Lecturio course “Emergency Medicine” ▻ WATCH the complete course on ...
Lecturio Medical
Biocompatibility: Applying the New ISO 10993 Standards
A new updated ISO 10993-1 standard came out in Aug of 2018 that drastically changed how we access medical devices for biocomaptibility. We also are ...
Nelson Labs
Public Workshop: A Framework for Regulatory Use of Real-World Evidence
Duke Margolis
How to Use Biocompatibility to Evaluate Changes in a Medical Device
Medical devices often go through many changes throughout their normal product lifecycles. Whenever a device goes through one of these changes, it should ...
Nelson Labs
ICH Q3D 'Elemental Impurities' and M7 'Mutagenic impurities' -- recent considerations
Speaker: Diana Van Riet, Medicines Evaluation Board (MEB), The Netherlands Session 1: Building quality documentation early during development ICH Q3D ...
emainfo
Preventing Adverse Health Effects from Nanotechnology
From smartphones to skincare, there are currently over 1000 commercial products containing nanomaterials, with applications as far ranging as the fields of ...
Centers for Disease Control and Prevention (CDC)
Biocompatibility Testing of Combination Products & Medical Devices: New Regulatory Guidance
Learn how to implement an efficient testing program for assessing the biocompatibility of a combination product's device component to satisfy both US and ...
Eurofins Medical Device Testing
Principles of Medical Equipment Maintenance Automation
The 75-minute webinar will feature Mark Miller, a registered professional engineer, and Vice President of Engineering at Datrend Systems Inc. Mark has more ...
MD Publishing, Inc.
Changing a colorant in an approved medical device, what should I know?
Change is inevitable with medical devices, suppliers, materials, and locations. All these can have an impact in the safety of the device. However, one of the more ...
Nelson Labs
What's new with EU MDR and IVDR - TOPRA Symposium 2019
I decided to create a documentary of my visit to TOPRA Symposium 2019 in Dublin (October 1st, 2nd 2019) where I met so many people and guess what we ...
Easy Medical Device
Journey to safer chemicals - REACH registration
For 10 years, chemical companies have assessed the risks of their chemicals used in Europe and sent that information to the European Chemicals Agency ...
EUchemicals
Elastomeric Respirators for U.S. Healthcare Delivery
This webinar provides an overview of respiratory protection and guidance surrounding supply shortages. This webinar also provides information on infection ...
Centers for Disease Control and Prevention (CDC)
SME Assist: Basics of therapeutic goods regulation
This video explains the foundations of therapeutic goods regulation in Australia for Small and Medium Enterprises (SMEs) in the therapeutic goods industry.
TGA Australia
Develop a Biological Evaluation Plan (BEP)
The new FDA guidance document for ISO 10993-1 focuses on a risk based approach to biocompatibility. The first step is to develop a BEP. This webinar will go ...
Nelson Labs
Medical Oncologist Dr. Yelena Novik Discusses Drug Development Trials and Regulations
NYU Langone medical oncologist Dr. Yelena Novik offers a history of clinical trials and how they come to be regulated to protect people. Learn more about Dr.
NYU Langone Health
Drug discovery and development process
Discovering and bringing one new drug to the market typically takes an average of 14 years of research and clinical development efforts. Learn about the many ...
Novartis
Importing and Exporting CBER Regulated Products
This video describes the U.S Food and Drug Administration Center for Biologics Evaluation and Research's role in regulating the import and export of biological ...
Centers for Disease Control and Prevention (CDC)
Assessment And Screening Co-occurring Disorders-1.m4v
Training CEUs for Licensed Professional Counselors (LPC) and Licensed Mental Health Counselors (LMHC) by AllCEUs.
AllCEUs Counseling Education
The Power of Extractable/Leachable Chemistry Testing for Medical Devices
This webinar summarizes how extractable/leachable chemistry testing for medical devices can save time and money for manufacturers preparing for a 510(k) ...
Nelson Labs
EMT 2-1: Scene Size-Up
Module 2-1 of the Wisconsin EMT curriculum - Scene Size-Up.
WCTCEMS
JECFA. Evaluating the safety of food additives.
JECFA is an international scientific expert committee administered jointly by the Food and Agriculture Organization of the United Nations (FAO) and WHO.
World Health Organization (WHO)
When Alcohol, Summer, COVID-19 and Teens Intersect
At Caron, we understand that different people have different needs in treatment. Learn more about our inpatient programs here Teens: ...
CaronTreatment